As a clinician, I was pleased to learn about ZILBRYSQ as a treatment option for my adult patients with gMG. This FDA-approved self-administered complement C5 inhibitor is approved for treating anti-AChR Ab+ gMG in adults. I encourage you to explore the clinical data to determine how it may benefit your practice.



As a clinician, I was pleased to learn about ZILBRYSQ as a treatment option for my adult patients with gMG. This FDA-approved self-administered complement C5 inhibitor is approved for treating anti-AChR Ab+ gMG in adults. I encourage you to explore the clinical data to determine how it may benefit your practice.
Dr Suraj Muley is a paid consultant of UCB, Inc.
Suraj Muley, MD*
Professor of Neurology
Bob Bove Neuroscience Institute
Paradise Valley, AZ
ZILBRYSQ is a first-of-its-kind small peptide C5 inhibitor approved for the treatment of gMG in adult patients who are anti-AChR Ab+1,2
STUDY DESIGNS
ZILBRYSQ was studied in a Phase 3 pivotal trial and extension study
RAISE: Phase 3 study included adult patients with mild-to-severe anti-AChR Ab+ gMG1
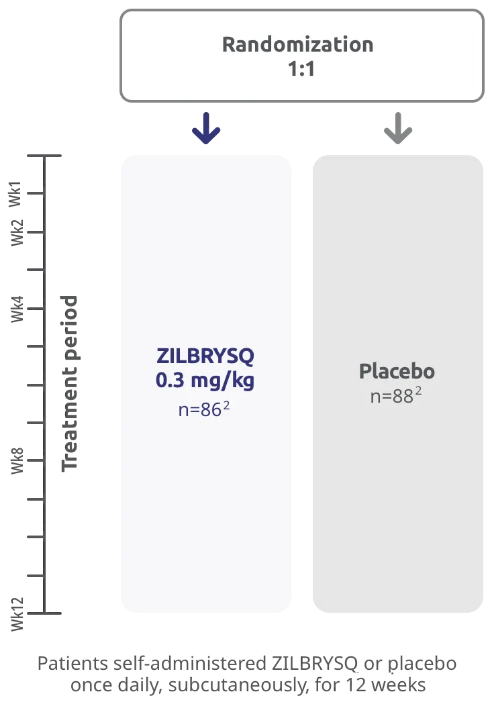
The efficacy and safety of ZILBRYSQ for the treatment of generalized myasthenia gravis (gMG) in adult patients who are anti-acetylcholine receptor (AChR) antibody positive (Ab+) were established in a 12-week, multicenter, randomized, double-blind, placebo-controlled study.
A total of 174 patients were randomized to receive either ZILBRYSQ (n=86) or placebo (n=88) once daily by subcutaneous injection. Patients on concomitant medications to treat gMG continued on therapy at stable doses throughout the study.
RAISE study design


Primary Endpoint at Week 12:
- Change from baseline (CFB) in MG-Activities of Daily Living (MG-ADL) total score
Select Secondary Endpoints at Week 12:
- CFB in Quantitative Myasthenia Gravis (QMG) score
- CFB in Myasthenia Gravis Composite (MGC) score
- CFB in Myasthenia Gravis Quality of Life-15 revised (MG-QoL 15r) score
Inclusion criteria2
- Diagnosis of mild-to-severe gMG (Myasthenia Gravis Foundation of America clinical classification class II to IV)
- Positive serology for AChR-binding autoantibodies
- MG-ADL total score of ≥6
- Those on MG therapy prior to screening needed to maintain a stable dose (includes acetylcholinesterase [AChE] inhibitors, steroids, or non-steroidal immunosuppressive therapies [NSISTs], either in combination or alone)
Exclusion criteria included1:
- Those who had had a thymectomy within 12 months before baseline or were scheduled to have one during the 12-week study
- Those who had received treatment with intravenous immunoglobulin, subcutaneous immunoglobulin, or plasma exchange within 4 weeks before baseline
Patient demographics1
| Placebo (n=88) |
ZILBRYSQ 0.3 mg/kg (n=86) |
|
|---|---|---|
| Mean age in years (SD) | 53.3 (15.7) | 52.6 (14.6) |
| Gender distribution, female, n (%) | 47 (53%) | 52 (60%) |
| Race, White, n (%) | 62 (70%) | 66 (77%) |
| Race, Asian, n (%) | 14 (16%) | 7 (8%) |
| Race, Black, n (%) | 7 (8%) | 6 (7%) |
| Race, American Indian or Alaskan native, n (%) | 1 (1%) | 0 |
| Race, missing, n (%) | 4 (5%) | 7 (8%) |
| Ethnicity, Hispanic or Latino, n (%) | 5 (6%) | 7 (8%) |
| Ethnicity, missing, n (%) | 4 (5%) | 7 (8%) |
| Mean duration of disease in years (SD) | 9.0 (10.4) | 9.3 (9.5) |
| MGFA class II, n (%) | 27 (31%) | 22 (26%) |
| MGFA class III, n (%) | 57 (65%) | 60 (70%) |
| MGFA class IV, n (%) | 4 (5%) | 4 (5%) |
| Mean MG-ADL score (SD) | 10.9 (3.4) | 10.3 (2.5) |
| Mean QMG score (SD) | 19.4 (4.5) | 18.7 (3.6) |
| Mean MGC score (SD) | 21.6 (7.2) | 20.1 (6.0) |
| Mean MG-QoL 15r score (SD) | 18.9 (6.8) | 18.6 (6.6) |
| Treatment refractory, n (%)* | 44 (50%) | 44 (51%) |
| Baseline myasthenia gravis medications, n (%)† | ||
| Cholinesterase inhibitor | 73 (83%) | 74 (86%) |
| Corticosteroids | 51 (58%) | 59 (69%) |
| Azathioprine or mycophenolate mofetil | 35 (40%) | 30 (35%) |
| Cyclosporine, methotrexate, or tacrolimus | 15 (17%) | 12 (14%) |
A participant was considered treatment refractory if they had treatment for at least 1 year with 2 or more of the following therapies: prednisone, azathioprine, mycophenolate, cyclosporine, cyclophosphamide, methotrexate, tacrolimus, rituximab, eculizumab, or other corticosteroids; or a history of treatment with at least 1 of these therapies for 1 year or more and required chronic plasma exchange, intravenous immunoglobulin, or subcutaneous immunoglobulin at least every 3 months for the 12 months before enrollment.
Safety set, includes all participants who received at least 1 dose of study drug with participants analyzed based on the study treatment received.
RAISE-XT: an extension study of adult patients who opted to continue on ZILBRYSQ or switch to ZILBRYSQ from placebo3,4
The primary objective of RAISE-XT was to evaluate the long-term safety and tolerability of ZILBRYSQ in study participants with gMG. Long-term efficacy was also studied through multiple measures as select secondary endpoints. This open-label extension is an ongoing study with the current interim data cut (November 11, 2023) at Week E108.
RAISE-XT: extension study‡
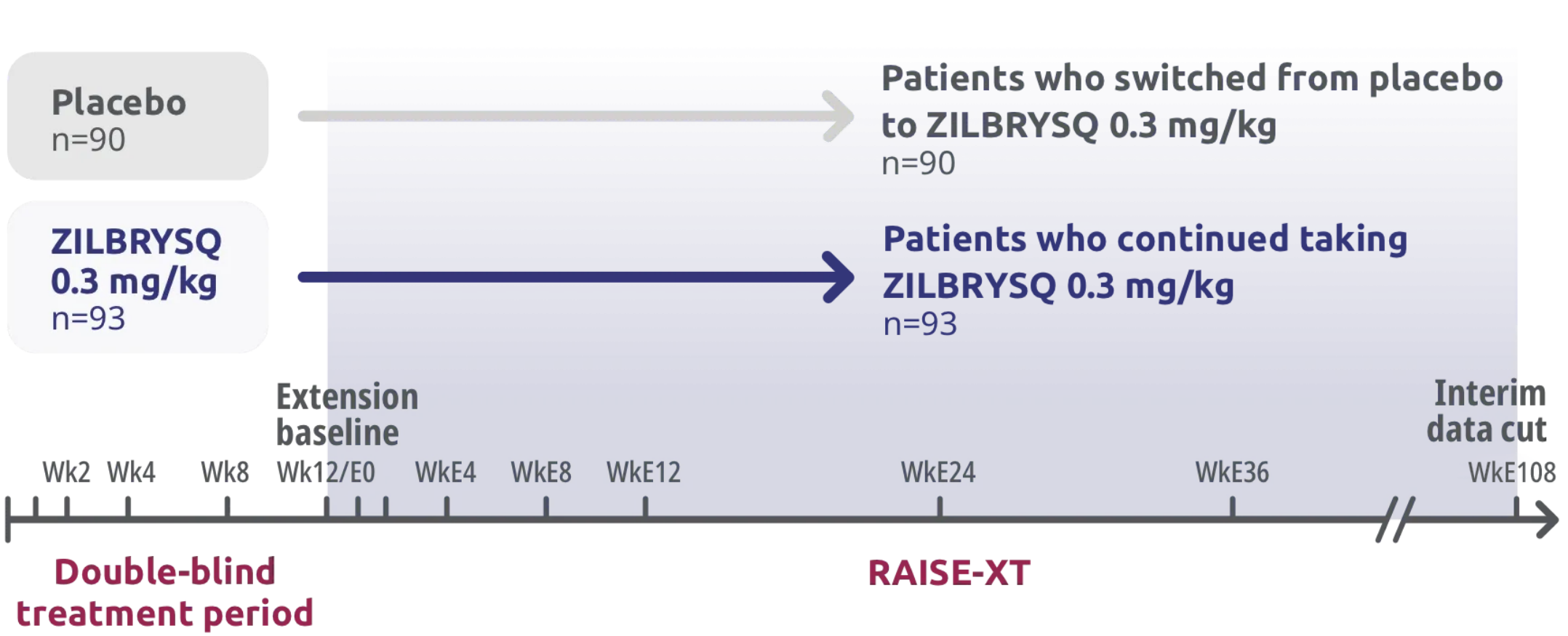

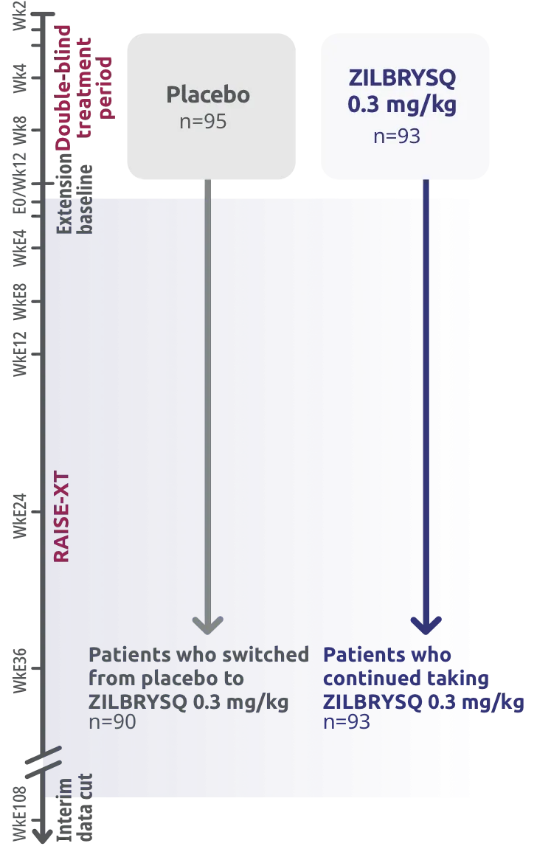
RAISE-XT evaluated 200 adult patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy.3,4
At the time of data cutoff (November 11, 2023), most patients who entered RAISE-XT (73%) were still enrolled, with no discontinuations reported by the investigators as being due to lack of efficacy.5
Primary Safety Endpoint:
- Incidence of treatment-emergent adverse events
100% of patients who completed the RAISE study opted into RAISE-XT.3
Select Secondary Endpoints at Week E12:
- CFB in MG-ADL score
- CFB in QMG score
- CFB in MGC score
- CFB in MG-QoL 15r score
EFFICACY
RAISE: Primary Endpoint MG-ADL
Rapid and statistically significant improvements in patients’ ability to manage activities of daily living at Week 121,2
Primary endpoint: CFB in MG-ADL total score at Week 12


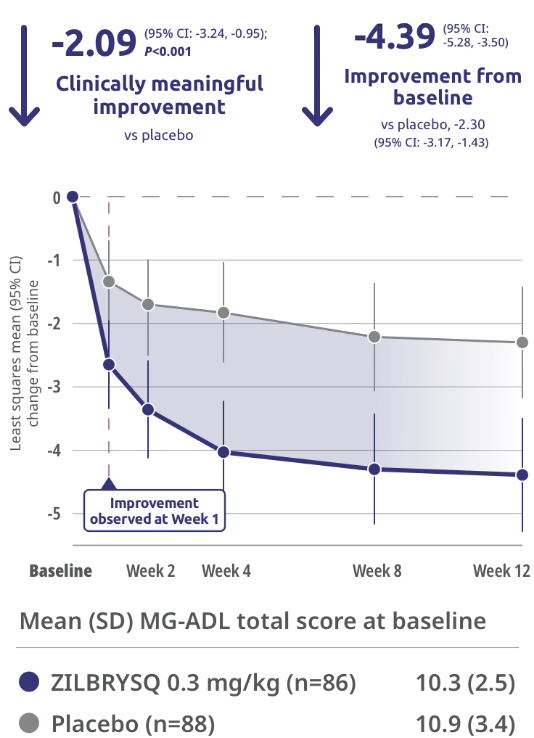
Clinically meaningful was defined as a ≥2-point change in MG-ADL score.1
The safety and efficacy of ZILBRYSQ were evaluated in a 12-week, multicenter, randomized, double-blind, placebo-controlled study. The patient population included adult patients with a diagnosis of mild-to-severe gMG (MGFA class II-IV).2
The most common adverse reactions (reported in at least 10% of patients treated with ZILBRYSQ) were injection site reactions, upper respiratory tract infections, and diarrhea.2
MG-ADL
Assesses the impact of gMG on daily functions of 8 symptoms on a scale of 0 to 3, with total scores ranging from 0 to 24. Higher scores are interpreted as greater impairments.2
- An improvement of ≥2 points was established as clinically meaningful.1
Measures include6:
- Talking
- Chewing
- Swallowing
- Breathing
- Brushing teeth and/or combing hair
- Rising from a chair
- Diplopia
- Eyelid droop
RAISE-XT: Secondary Endpoint MG-ADL
Sustained efficacy through Week E1083,5
The primary endpoint of RAISE-XT evaluated the long-term safety and tolerability of ZILBRYSQ.
Secondary endpoint: 3.69-point CFB observed at Week E12 in MG-ADL score for patients who switched to ZILBRYSQ from placebo (mITT population)
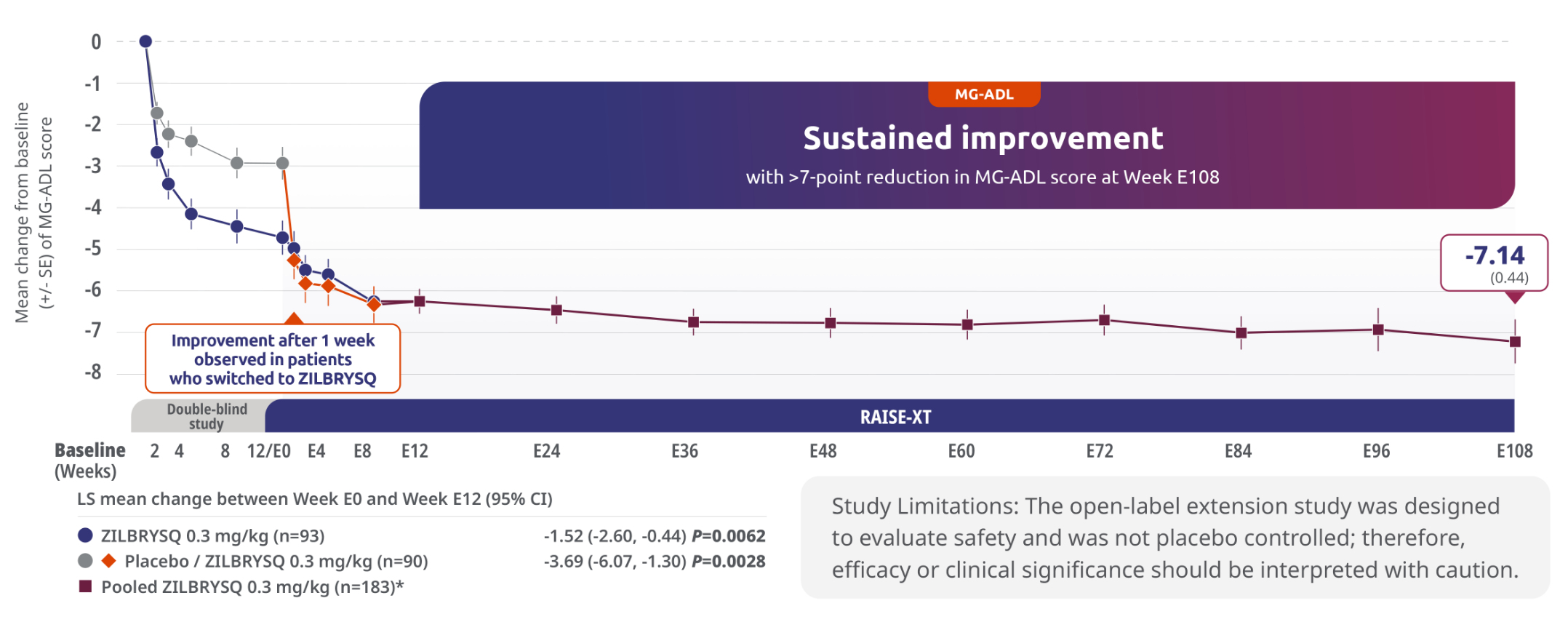
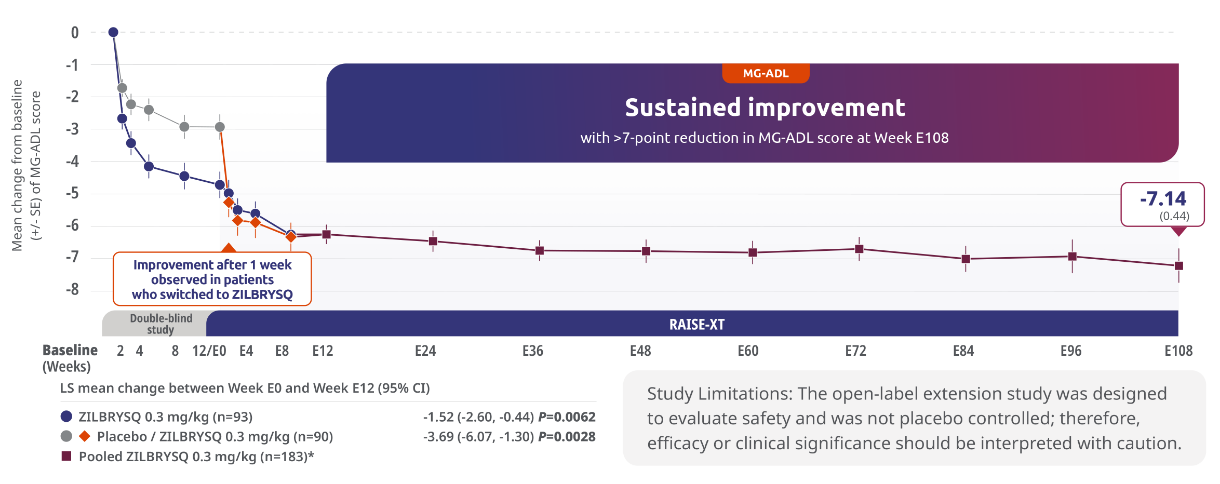
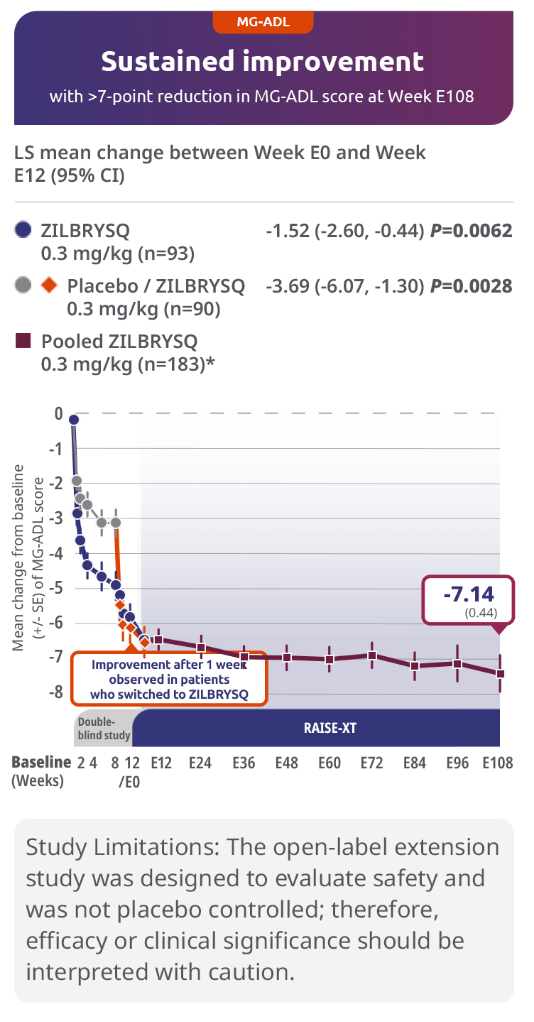
The long-term safety, tolerability, and efficacy were evaluated in an open-label extension study that comprised gMG patients who participated in either Phase 2 or Phase 3 (RAISE) of the clinical trials.3
RAISE-XT evaluated 200 adult patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the 183 patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy. Data cutoff: November 11, 2023.3,4
Includes pooled data of patients from Week E12-Week E108.4
MG-ADL responder rates for ZILBRYSQ in RAISE and RAISE-XT2,4
A high proportion of patients taking ZILBRYSQ were MG-ADL clinical responders (≥3-point improvement from baseline) at Week 12 and Week E108.2,4

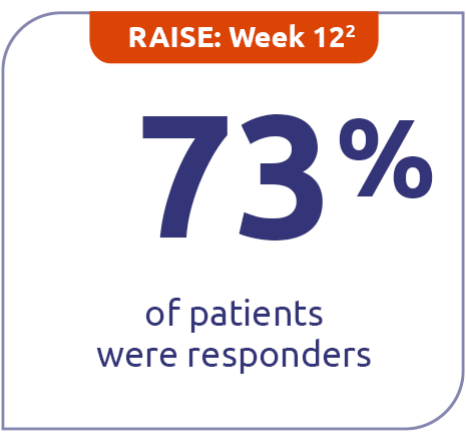
73% (n=86) with ZILBRYSQ in
RAISE vs 46% (n=88) with placebo
(P<0.001)2
73% (n=86) with ZILBRYSQ in RAISE vs 46% (n=88) with placebo (P<0.001)2

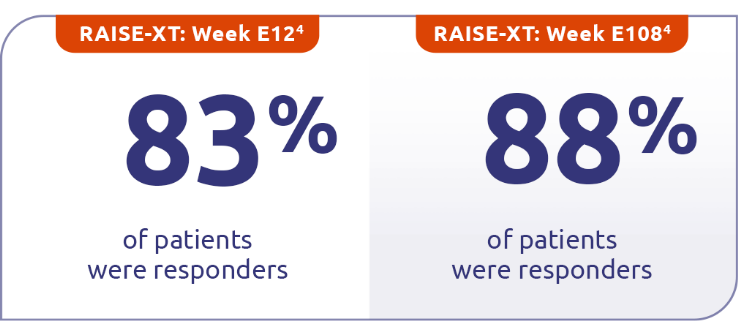
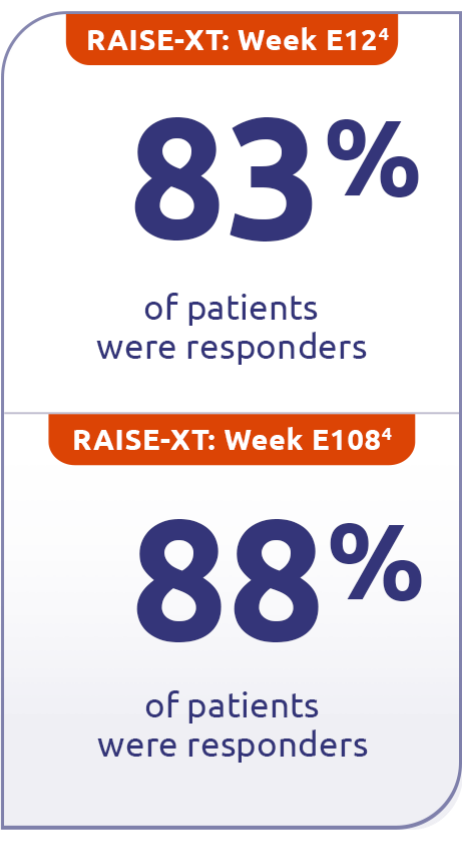
In pooled data of patients taking ZILBRYSQ in RAISE-XT (n=183), 83% were responders at Week E12, followed by 88% at Week E1084†
Responder rate was specified as an other secondary efficacy endpoint in the RAISE study and an exploratory efficacy endpoint in RAISE-XT. Results should be interpreted with caution.1,3
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
Includes pooled data of patients from Phases 2 and 3 of the clinical study who continued on ZILBRYSQ or switched to ZILBRYSQ from placebo. Data cutoff: November 11, 2023.4
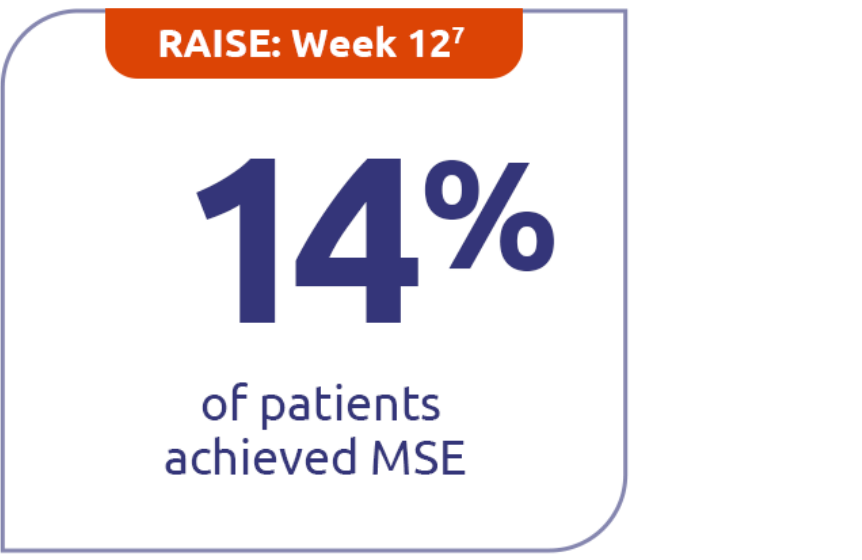
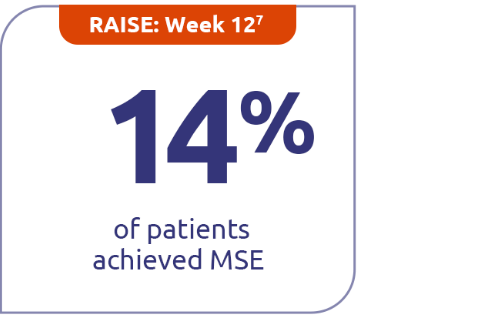
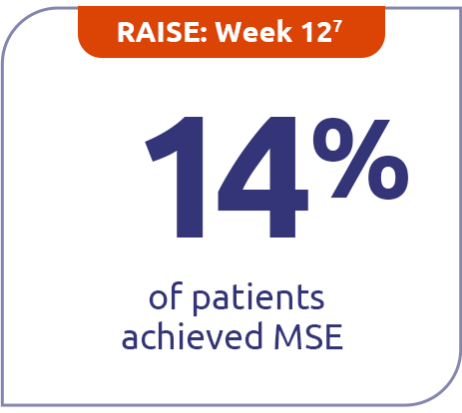
Continued minimal symptom expression (MSE) in patients through Week E1084
Patients who achieved MSE (mITT population) in either RAISE or RAISE-XT achieved an MG-ADL score of 0 or 1 without rescue therapy.3
Mean (SD) MG-ADL total
score at baseline1
- 10.9 (3.4) for placebo
- 10.3 (2.5) for ZILBRYSQ
14% (n=86) with ZILBRYSQ in RAISE vs 5.8% (n=88) with placebo (P=0.0885)7
14% (n=86) with ZILBRYSQ in RAISE vs 5.8% (n=88) with placebo (P=0.0885)7


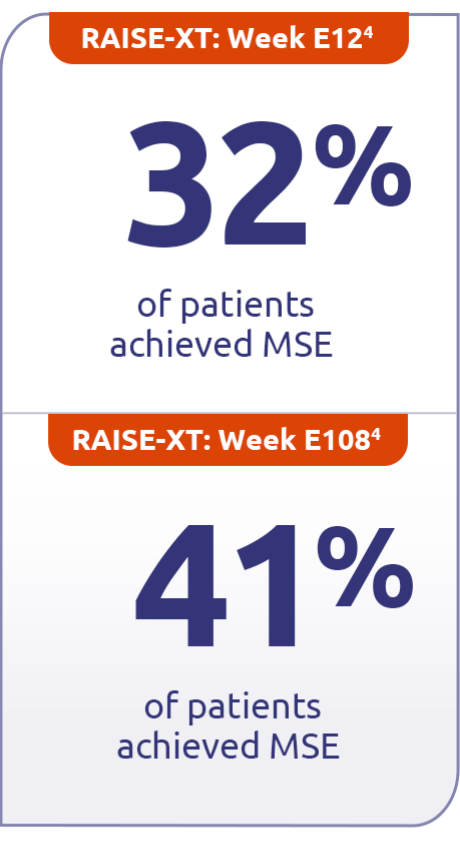
Mean (SD) MG-ADL total score at baseline1
- 6.3 (4.3) for ZILBRYSQ4
In pooled data of patients taking ZILBRYSQ in RAISE-XT (n=183)4‡
MSE was defined as an MG-ADL total score of 0-1. MSE was specified as an other secondary efficacy endpoint in the RAISE study and an exploratory endpoint in RAISE-XT. Results should be interpreted with caution.1,3
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
Includes pooled data of patients from Week E12-Week E108.4
RAISE-XT evaluated 200 adult patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the 183 patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy. Data cutoff: November 11, 2023.3,4
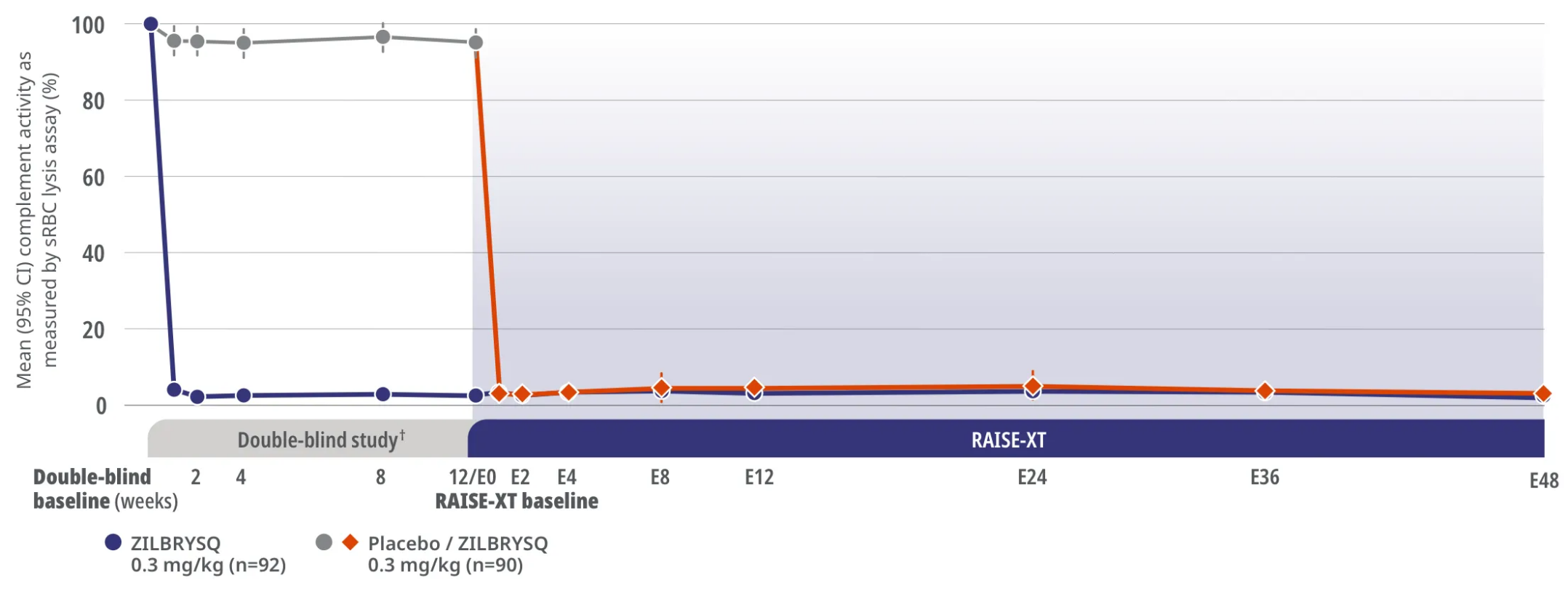
RAISE-XT Complement Inhibition
Complement inhibition was achieved after Week 1 and sustained through Week E483*
RAISE XT Other Endpoint: Complement activity as shown by sRBC lysis assay

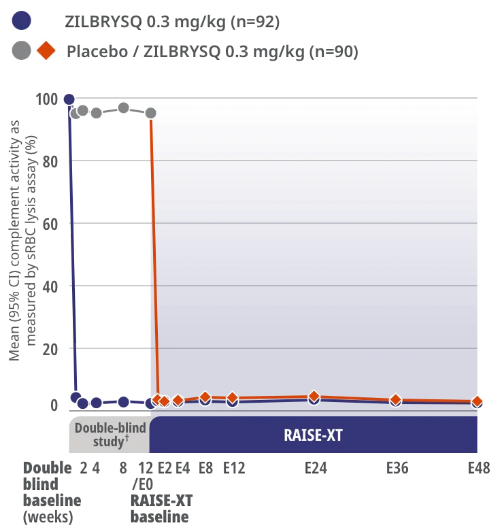
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
Assessment of complement activity using sRBC lysis assay was a pharmacodynamics outcome.3
In RAISE-XT, Week E48 was the data cut (September 8, 2022) for the assessment of complement activity.3
RAISE-XT is an ongoing, multicenter, open-label extension study of ZILBRYSQ in adult study participants with anti-AChR Ab+ gMG who completed the Phase 2 or Phase 3 (RAISE) study (N=200). The analysis cutoff date was September 8, 2022.3
The safety and efficacy of ZILBRYSQ were evaluated in RAISE, a 12-week, multicenter, randomized, double-blind, placebo-controlled study. The patient population included adult patients with a diagnosis of mild-to-severe gMG (MGFA class II-IV).2
CI=confidence interval; LS=least squares; MGFA=Myasthenia Gravis Foundation of America; mITT=modified intention-to-treat; SD=standard deviation; SE=standard error; sRBC=sheep red blood cell.
References:
- Howard JF Jr, Bresch S, Genge A, et al; RAISE Study Team. Safety and efficacy of zilucoplan in patients with generalised myasthenia gravis (RAISE): a randomised, double-blind, placebo-controlled, Phase 3 study. Lancet Neurol. 2023;22(5):395-406. doi:10.1016/S1474-4422(23)00080-7
- ZILBRYSQ [Prescribing Information]. Smyrna, GA: UCB, Inc.
- Howard JF Jr, Bresch S, Farmakidis C, et al. Long-term safety and efficacy of zilucoplan in patients with generalized myasthenia gravis: interim analysis of the RAISE-XT open-label extension study. Ther Adv Neurol Disord. 2024;17(3):1-16. doi:10.1177/17562864241243186
- Howard JF Jr, Freimer M, Genge A, et al. Response rates with zilucoplan in generalised myasthenia gravis: 120-week interim analysis of RAISE-XT. Presented at: International Congress on Neuromuscular Diseases; October 25-29, 2024; Perth, Australia. Session OS.03.06.
- Howard JF Jr, Freimer M, Genge A, et al; on behalf of the RAISE-XT Study Team. Long-term safety and efficacy of zilucoplan in generalized myasthenia gravis: 120-week interim analysis of RAISE-XT. Poster presented at: American Academy of Neuromuscular & Electrodiagnostic Medicine Annual Meeting; October 15-18, 2024; Savannah, GA. Poster 192.
- Conquer MG. MG Activities of Daily Living (MG-ADL) scale. September 29, 2022. Accessed November 9, 2023. https://www.myastheniagravis.org/mg-activities-of-daily-living-mg-adl-scale/
- Vu T, Genge A, Hussain Y, et al; on behalf of the RAISE Investigators. Efficacy and safety of zilucoplan in myasthenia gravis: responder analysis from the randomized Phase 3 RAISE trial. Poster presented at: American Association of Neuromuscular & Electrodiagnostic Medicine Annual Meeting; September 21-24, 2022; Nashville, TN. Poster 200.














